Search
- Page Path
- HOME > Search
Original Article
- Thyroid
- Lobeglitazone, A Peroxisome Proliferator-Activated Receptor-Gamma Agonist, Inhibits Papillary Thyroid Cancer Cell Migration and Invasion by Suppressing p38 MAPK Signaling Pathway
- Jun-Qing Jin, Jeong-Sun Han, Jeonghoon Ha, Han-Sang Baek, Dong-Jun Lim
- Endocrinol Metab. 2021;36(5):1095-1110. Published online October 14, 2021
- DOI: https://doi.org/10.3803/EnM.2021.1155
- 4,759 View
- 159 Download
- 9 Web of Science
- 8 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Background
Peroxisome proliferator-activated receptor-gamma (PPAR-γ) ligands have been widely shown to correlate with epithelial-mesenchymal transition (EMT) and cancer progression. Lobeglitazone (LGZ) is a novel ligand of PPAR-γ; and its role in EMT and metastasis in papillary thyroid carcinoma (PTC) is poorly understood. We aimed to investigate the role of LGZ in metastatic behavior of PTC cells.
Methods
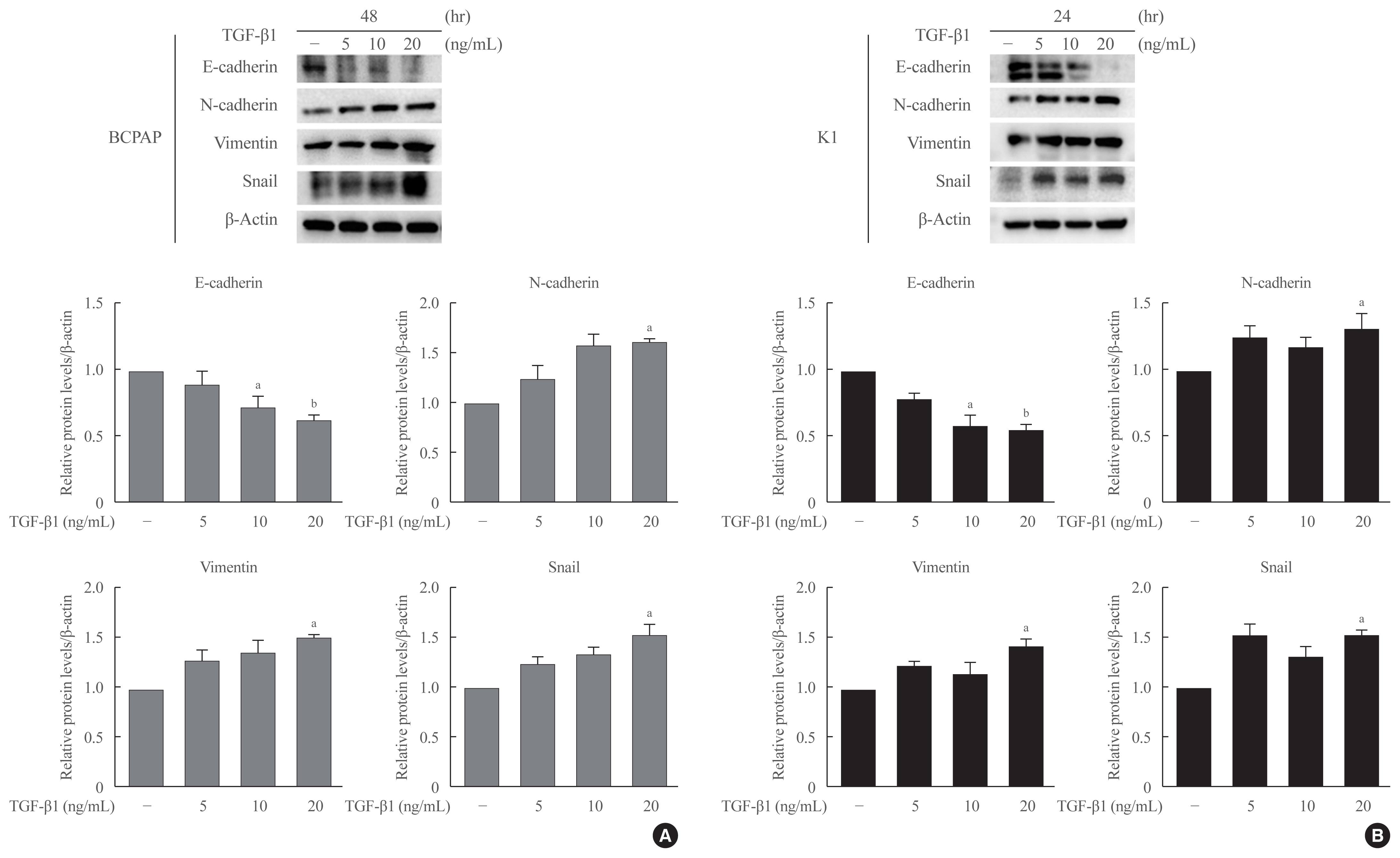
Half maximal inhibitory concentration (IC50) values of LGZ in BRAF-mutated PTC cell lines (BCPAP and K1) were determined using MTT assay. Rosiglitazone (RGZ), the PPAR-γ ligand was used as a positive control. The protein expression of PPAR-γ, cell-surface proteins (E-cadherin, N-cadherin), cytoskeletal protein (Vimentin), transcription factor (Snail), p38 mitogenactivated protein kinase (MAPK), extracellular signal-regulated kinase (ERK) 1/2 pathway, and matrix metalloproteinase (MMP)-2 expression were measured using Western blotting. Changes in E-cadherin expression were also determined using immunocytochemistry. Cell migration and invasion were analyzed using wound healing and Matrigel invasion assays.
Results
Treatment with LGZ or RGZ significantly inhibited transforming growth factor-beta1 (TGF-β1)-induced EMT-associated processes such as fibroblast-like morphological changes, EMT-related protein expression, and increased cell migration and invasion in BCPAP and K1 cells. LGZ restored TGF-β1-induced loss of E-cadherin, as observed using immunocytochemistry. Furthermore, LGZ and RGZ suppressed TGF-β1-induced MMP-2 expression and phosphorylation of p38 MAPK, but not ERK1/2. Although there was no change in PPAR-γ expression after treatment with LGZ or RGZ, the effect of downstream processes mediated by LGZ was hampered by GW9662, a PPAR-γ antagonist.
Conclusion
LGZ inhibits TGF-β1-induced EMT, migration, and invasion through the p38 MAPK signaling pathway in a PPAR-γ-dependent manner in PTC cells. -
Citations
Citations to this article as recorded by- Diabetes Mellitus and Thyroid Cancers: Risky Correlation, Underlying Mechanisms and Clinical Prevention
Rongqian Wu, Junping Zhang, Guilin Zou, Shanshan Li, Jinying Wang, Xiaoxinlei Li, Jixiong Xu
Diabetes, Metabolic Syndrome and Obesity.2024; Volume 17: 809. CrossRef - Clinicopathological Evaluation of Papillary Thyroid Microcarcinoma
Ando Takahito, Kimihito Fujii, Hirona Banno, Masayuki Saito, Yukie Ito, Mirai Ido, Manami Goto, Yukako Mouri, Junko Kousaka, Tsuneo Imai, Shogo Nakano
Cureus.2024;[Epub] CrossRef - Pioglitazone, a peroxisome proliferator‑activated receptor γ agonist, induces cell death and inhibits the proliferation of hypoxic HepG2 cells by promoting excessive production of reactive oxygen species
Guohao Huang, Mengfan Zhang, Manzhou Wang, Wenze Xu, Xuhua Duan, Xinwei Han, Jianzhuang Ren
Oncology Letters.2024;[Epub] CrossRef - The Activation of PPARγ by (2Z,4E,6E)-2-methoxyocta-2,4,6-trienoic Acid Counteracts the Epithelial–Mesenchymal Transition Process in Skin Carcinogenesis
Enrica Flori, Sarah Mosca, Giorgia Cardinali, Stefania Briganti, Monica Ottaviani, Daniela Kovacs, Isabella Manni, Mauro Truglio, Arianna Mastrofrancesco, Marco Zaccarini, Carlo Cota, Giulia Piaggio, Mauro Picardo
Cells.2023; 12(7): 1007. CrossRef - Cumulative exposure to metabolic syndrome increases thyroid cancer risk in young adults: a population-based cohort study
Jinyoung Kim, Kyungdo Han, Mee Kyoung Kim, Ki-Hyun Baek, Ki-Ho Song, Hyuk-Sang Kwon
The Korean Journal of Internal Medicine.2023; 38(4): 526. CrossRef - Drug repositioning in thyroid cancer treatment: the intriguing case of anti-diabetic drugs
Alessia Greco, Francesca Coperchini, Laura Croce, Flavia Magri, Marsida Teliti, Mario Rotondi
Frontiers in Pharmacology.2023;[Epub] CrossRef - Fish and the Thyroid: A Janus Bifrons Relationship Caused by Pollutants and the Omega-3 Polyunsaturated Fatty Acids
Salvatore Benvenga, Fausto Famà, Laura Giovanna Perdichizzi, Alessandro Antonelli, Gabriela Brenta, Francesco Vermiglio, Mariacarla Moleti
Frontiers in Endocrinology.2022;[Epub] CrossRef - Identifying and categorizing compounds that reduce corneal transforming growth factor beta induced protein levels: a scoping review
Gabriella Guo Sciriha, Janet Sultana, Joseph Borg
Expert Review of Clinical Pharmacology.2022; 15(12): 1423. CrossRef
- Diabetes Mellitus and Thyroid Cancers: Risky Correlation, Underlying Mechanisms and Clinical Prevention


 KES
KES

 First
First Prev
Prev



